Kolekce Atom Is Neutral If Zdarma
Kolekce Atom Is Neutral If Zdarma. Neutral atoms of an element contain an equal number of protons and electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. Sometimes atoms are more stable though when they are not perfectly electrically neutral. The number of protons determines an element's atomic number (z) and distinguishes one element from another.
Prezentováno Cation Vs Anion Definition Chart And The Periodic Table Technology Networks
When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. This can cause atoms to take on a temporary charge from a nearby atom. Atoms which are not electrically neutral are called ions.Why is an atom …
This means that it has an equal number if protons and electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. A normal atom has a neutral charge with equal numbers of positive and negative particles. Neutral atoms of an element contain an equal number of protons and electrons. Some atoms can attract additional electrons so they become negatively charged. The number of protons determines an element's atomic number (z) and distinguishes one element from another.

Ions are atoms with extra electrons or missing electrons.. They contain the same number of protons as electrons. Moreover neutrons are neural and do not cause any change. A normal atom has a neutral charge with equal numbers of positive and negative particles. Atoms which are not electrically neutral are called ions. If it does then the charges cancel each other out, making a neutral atom. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. This can cause atoms to take on a temporary charge from a nearby atom. For example, carbon's atomic number (z) is 6 because it has 6 protons.. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another.

Why is an atom …. They contain the same number of protons as electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. Sometimes atoms are more stable though when they are not perfectly electrically neutral. Atoms which are not electrically neutral are called ions. If there is a … That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number.

A normal atom has a neutral charge with equal numbers of positive and negative particles. A neutral sodium atom, for example,. Ions are atoms with extra electrons or missing electrons. If it does then the charges cancel each other out, making a neutral atom. Neutral atoms of an element contain an equal number of protons and electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another... Moreover neutrons are neural and do not cause any change.

Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Moreover neutrons are neural and do not cause any change. For example, carbon's atomic number (z) is 6 because it has 6 protons. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. This means that it has an equal number if protons and electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. They contain the same number of protons as electrons. This means that the two atoms polarize each other:. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral.

… when an ion is formed, the number of protons does not change... Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. For example, carbon's atomic number (z) is 6 because it has 6 protons. Neutral atoms can be turned into positively charged ions by removing one or more electrons. A neutral sodium atom, for example,. Moreover neutrons are neural and do not cause any change. If there is a … For example, carbon's atomic number (z) is 6 because it has 6 protons. A normal atom has a neutral charge with equal numbers of positive and negative particles. Why is an atom …. Moreover neutrons are neural and do not cause any change.

Atoms which are not electrically neutral are called ions.. This can cause atoms to take on a temporary charge from a nearby atom. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. A neutral sodium atom, for example,. For example, carbon's atomic number (z) is 6 because it has 6 protons. If there is a … A normal atom has a neutral charge with equal numbers of positive and negative particles. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. When one or more electrons is stripped away from an atom, it becomes positively charged.
If it does then the charges cancel each other out, making a neutral atom.. … when an ion is formed, the number of protons does not change. Neutral atoms of an element contain an equal number of protons and electrons. A neutral sodium atom, for example,. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. This can cause atoms to take on a temporary charge from a nearby atom. A normal atom has a neutral charge with equal numbers of positive and negative particles. The number of protons determines an element's atomic number (z) and distinguishes one element from another.. This means that the two atoms polarize each other:

Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Ions are atoms with extra electrons or missing electrons. … when an ion is formed, the number of protons does not change. They contain the same number of protons as electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. If there is a … Neutral atoms of an element contain an equal number of protons and electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way.. They contain the same number of protons as electrons.

Neutral atoms of an element contain an equal number of protons and electrons... As long as the numbers of electron and protons are the same, the charges will balance. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. When one or more electrons is stripped away from an atom, it becomes positively charged. The number of protons determines an element's atomic number (z) and distinguishes one element from another. If it does then the charges cancel each other out, making a neutral atom. For example, carbon's atomic number (z) is 6 because it has 6 protons.. The number of protons determines an element's atomic number (z) and distinguishes one element from another.

The number of protons determines an element's atomic number (z) and distinguishes one element from another.. Atoms which are not electrically neutral are called ions. Some atoms can attract additional electrons so they become negatively charged. Neutral atoms can be turned into positively charged ions by removing one or more electrons. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. As long as the numbers of electron and protons are the same, the charges will balance. The number of protons determines an element's atomic number (z) and distinguishes one element from another. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. This means that the two atoms polarize each other:

For example, carbon's atomic number (z) is 6 because it has 6 protons. .. Neutral atoms can be turned into positively charged ions by removing one or more electrons.

This means that the two atoms polarize each other:.. A normal atom has a neutral charge with equal numbers of positive and negative particles. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. This can cause atoms to take on a temporary charge from a nearby atom. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. When one or more electrons is stripped away from an atom, it becomes positively charged. This means that it has an equal number if protons and electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. Why is an atom … Neutral atoms of an element contain an equal number of protons and electrons.. This means that the two atoms polarize each other:

The number of protons determines an element's atomic number (z) and distinguishes one element from another... This can cause atoms to take on a temporary charge from a nearby atom. Why is an atom … When one or more electrons is stripped away from an atom, it becomes positively charged. For example, carbon's atomic number (z) is 6 because it has 6 protons. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Ions are atoms with extra electrons or missing electrons. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. Sometimes atoms are more stable though when they are not perfectly electrically neutral. This means that the two atoms polarize each other: This means that it has an equal number if protons and electrons.. The number of protons determines an element's atomic number (z) and distinguishes one element from another.

This means that it has an equal number if protons and electrons. This can cause atoms to take on a temporary charge from a nearby atom. When one or more electrons is stripped away from an atom, it becomes positively charged. As long as the numbers of electron and protons are the same, the charges will balance. A normal atom has a neutral charge with equal numbers of positive and negative particles.

If it does then the charges cancel each other out, making a neutral atom. The number of protons determines an element's atomic number (z) and distinguishes one element from another. When one or more electrons is stripped away from an atom, it becomes positively charged. Moreover neutrons are neural and do not cause any change. Neutral atoms of an element contain an equal number of protons and electrons. Neutral atoms of an element contain an equal number of protons and electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another.

Some atoms can attract additional electrons so they become negatively charged... Neutral atoms can be turned into positively charged ions by removing one or more electrons. Neutral atoms of an element contain an equal number of protons and electrons. They contain the same number of protons as electrons. A normal atom has a neutral charge with equal numbers of positive and negative particles... As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another.

Neutral atoms of an element contain an equal number of protons and electrons. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. … when an ion is formed, the number of protons does not change. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. For example, carbon's atomic number (z) is 6 because it has 6 protons. This can cause atoms to take on a temporary charge from a nearby atom.. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number.

They contain the same number of protons as electrons.. A normal atom has a neutral charge with equal numbers of positive and negative particles. This can cause atoms to take on a temporary charge from a nearby atom. If there is a … Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. If it does then the charges cancel each other out, making a neutral atom. Why is an atom … Neutral atoms of an element contain an equal number of protons and electrons. As long as the numbers of electron and protons are the same, the charges will balance. The number of protons determines an element's atomic number (z) and distinguishes one element from another.. Sometimes atoms are more stable though when they are not perfectly electrically neutral.

That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. As long as the numbers of electron and protons are the same, the charges will balance. This means that the two atoms polarize each other: A normal atom has a neutral charge with equal numbers of positive and negative particles. If there is a … As long as the numbers of electron and protons are the same, the charges will balance.

Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. .. A normal atom has a neutral charge with equal numbers of positive and negative particles.

The number of protons determines an element's atomic number (z) and distinguishes one element from another... … when an ion is formed, the number of protons does not change. Atoms which are not electrically neutral are called ions. A normal atom has a neutral charge with equal numbers of positive and negative particles.. Ions are atoms with extra electrons or missing electrons.

For example, carbon's atomic number (z) is 6 because it has 6 protons... This can cause atoms to take on a temporary charge from a nearby atom.. This means that the two atoms polarize each other:

When one or more electrons is stripped away from an atom, it becomes positively charged. The number of protons determines an element's atomic number (z) and distinguishes one element from another. Why is an atom … The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. Sometimes atoms are more stable though when they are not perfectly electrically neutral. A normal atom has a neutral charge with equal numbers of positive and negative particles.. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number.

This means that it has an equal number if protons and electrons... When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. Moreover neutrons are neural and do not cause any change. This can cause atoms to take on a temporary charge from a nearby atom. As long as the numbers of electron and protons are the same, the charges will balance.. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral.

When one or more electrons is stripped away from an atom, it becomes positively charged.. … when an ion is formed, the number of protons does not change. They contain the same number of protons as electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. This means that it has an equal number if protons and electrons. If it does then the charges cancel each other out, making a neutral atom.. They contain the same number of protons as electrons.

… when an ion is formed, the number of protons does not change. Sometimes atoms are more stable though when they are not perfectly electrically neutral. For example, carbon's atomic number (z) is 6 because it has 6 protons. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Ions are atoms with extra electrons or missing electrons. Neutral atoms of an element contain an equal number of protons and electrons. As long as the numbers of electron and protons are the same, the charges will balance.

This means that the two atoms polarize each other: This can cause atoms to take on a temporary charge from a nearby atom... For example, carbon's atomic number (z) is 6 because it has 6 protons.

They contain the same number of protons as electrons. A normal atom has a neutral charge with equal numbers of positive and negative particles. Neutral atoms of an element contain an equal number of protons and electrons. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. Neutral atoms of an element contain an equal number of protons and electrons. Sometimes atoms are more stable though when they are not perfectly electrically neutral. Neutral atoms can be turned into positively charged ions by removing one or more electrons. When one or more electrons is stripped away from an atom, it becomes positively charged. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. If it does then the charges cancel each other out, making a neutral atom. They contain the same number of protons as electrons.

This means that it has an equal number if protons and electrons. When one or more electrons is stripped away from an atom, it becomes positively charged. For example, carbon's atomic number (z) is 6 because it has 6 protons. This means that the two atoms polarize each other: If there is a … This can cause atoms to take on a temporary charge from a nearby atom. Some atoms can attract additional electrons so they become negatively charged. As long as the numbers of electron and protons are the same, the charges will balance. Atoms which are not electrically neutral are called ions. Why is an atom … If it does then the charges cancel each other out, making a neutral atom... The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral.

Ions are atoms with extra electrons or missing electrons. Neutral atoms of an element contain an equal number of protons and electrons. Moreover neutrons are neural and do not cause any change. Neutral atoms of an element contain an equal number of protons and electrons.

This can cause atoms to take on a temporary charge from a nearby atom. A normal atom has a neutral charge with equal numbers of positive and negative particles. Neutral atoms of an element contain an equal number of protons and electrons. This means that the two atoms polarize each other: A neutral sodium atom, for example,. They contain the same number of protons as electrons. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. Some atoms can attract additional electrons so they become negatively charged. When one or more electrons is stripped away from an atom, it becomes positively charged. If there is a … That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. Ions are atoms with extra electrons or missing electrons.

As long as the numbers of electron and protons are the same, the charges will balance... The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral.. As long as the numbers of electron and protons are the same, the charges will balance.

When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way.. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral.. … when an ion is formed, the number of protons does not change.

Ions are atoms with extra electrons or missing electrons. Atoms which are not electrically neutral are called ions. For example, carbon's atomic number (z) is 6 because it has 6 protons. A normal atom has a neutral charge with equal numbers of positive and negative particles. Some atoms can attract additional electrons so they become negatively charged.

This can cause atoms to take on a temporary charge from a nearby atom. This means that it has an equal number if protons and electrons. As long as the numbers of electron and protons are the same, the charges will balance. A normal atom has a neutral charge with equal numbers of positive and negative particles. Why is an atom … This means that the two atoms polarize each other: Ions are atoms with extra electrons or missing electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. When one or more electrons is stripped away from an atom, it becomes positively charged... This means that it has an equal number if protons and electrons.

They contain the same number of protons as electrons... As long as the numbers of electron and protons are the same, the charges will balance. They contain the same number of protons as electrons. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. The number of protons determines an element's atomic number (z) and distinguishes one element from another. This can cause atoms to take on a temporary charge from a nearby atom. Moreover neutrons are neural and do not cause any change.. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral.

When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. If there is a … Neutral atoms of an element contain an equal number of protons and electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. A neutral sodium atom, for example,. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. This means that the two atoms polarize each other: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. This can cause atoms to take on a temporary charge from a nearby atom. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number.. For example, carbon's atomic number (z) is 6 because it has 6 protons.

This means that the two atoms polarize each other: Neutral atoms can be turned into positively charged ions by removing one or more electrons. Atoms which are not electrically neutral are called ions.. A normal atom has a neutral charge with equal numbers of positive and negative particles.

… when an ion is formed, the number of protons does not change. For example, carbon's atomic number (z) is 6 because it has 6 protons. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. A neutral sodium atom, for example,. Some atoms can attract additional electrons so they become negatively charged. If there is a … Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Neutral atoms of an element contain an equal number of protons and electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. For example, carbon's atomic number (z) is 6 because it has 6 protons. A normal atom has a neutral charge with equal numbers of positive and negative particles.. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges.

Some atoms can attract additional electrons so they become negatively charged.. This can cause atoms to take on a temporary charge from a nearby atom.. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number.

Neutral atoms can be turned into positively charged ions by removing one or more electrons.. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. A normal atom has a neutral charge with equal numbers of positive and negative particles. Moreover neutrons are neural and do not cause any change. If there is a … This means that the two atoms polarize each other: Sometimes atoms are more stable though when they are not perfectly electrically neutral. As long as the numbers of electron and protons are the same, the charges will balance. Ions are atoms with extra electrons or missing electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. Neutral atoms can be turned into positively charged ions by removing one or more electrons. They contain the same number of protons as electrons.
When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. For example, carbon's atomic number (z) is 6 because it has 6 protons. Ions are atoms with extra electrons or missing electrons. Moreover neutrons are neural and do not cause any change. A neutral sodium atom, for example,. A normal atom has a neutral charge with equal numbers of positive and negative particles. This means that the two atoms polarize each other: Sometimes atoms are more stable though when they are not perfectly electrically neutral... The number of protons determines an element's atomic number (z) and distinguishes one element from another.

This means that the two atoms polarize each other: That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Sometimes atoms are more stable though when they are not perfectly electrically neutral. Ions are atoms with extra electrons or missing electrons. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges.

When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. This can cause atoms to take on a temporary charge from a nearby atom. Ions are atoms with extra electrons or missing electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. When one or more electrons is stripped away from an atom, it becomes positively charged. Neutral atoms of an element contain an equal number of protons and electrons. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. This means that it has an equal number if protons and electrons. Why is an atom … This can cause atoms to take on a temporary charge from a nearby atom.

If there is a …. This means that it has an equal number if protons and electrons. As long as the numbers of electron and protons are the same, the charges will balance. Why is an atom … For example, carbon's atomic number (z) is 6 because it has 6 protons. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. When one or more electrons is stripped away from an atom, it becomes positively charged.
This can cause atoms to take on a temporary charge from a nearby atom... This means that the two atoms polarize each other: Neutral atoms of an element contain an equal number of protons and electrons. Sometimes atoms are more stable though when they are not perfectly electrically neutral. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Atoms which are not electrically neutral are called ions. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. The number of protons determines an element's atomic number (z) and distinguishes one element from another. If it does then the charges cancel each other out, making a neutral atom. Some atoms can attract additional electrons so they become negatively charged. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number.. Atoms which are not electrically neutral are called ions.
For example, carbon's atomic number (z) is 6 because it has 6 protons.. If it does then the charges cancel each other out, making a neutral atom. Atoms which are not electrically neutral are called ions... The number of protons determines an element's atomic number (z) and distinguishes one element from another.

If it does then the charges cancel each other out, making a neutral atom.. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. The number of protons determines an element's atomic number (z) and distinguishes one element from another. If it does then the charges cancel each other out, making a neutral atom. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral.

A neutral sodium atom, for example,. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Moreover neutrons are neural and do not cause any change. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another... A normal atom has a neutral charge with equal numbers of positive and negative particles.

Moreover neutrons are neural and do not cause any change... As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. Why is an atom … As long as the numbers of electron and protons are the same, the charges will balance. The number of protons determines an element's atomic number (z) and distinguishes one element from another. Neutral atoms of an element contain an equal number of protons and electrons. Atoms which are not electrically neutral are called ions. A neutral sodium atom, for example,. If it does then the charges cancel each other out, making a neutral atom... Why is an atom …

This can cause atoms to take on a temporary charge from a nearby atom. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. They contain the same number of protons as electrons. If it does then the charges cancel each other out, making a neutral atom. This means that it has an equal number if protons and electrons. When one or more electrons is stripped away from an atom, it becomes positively charged.
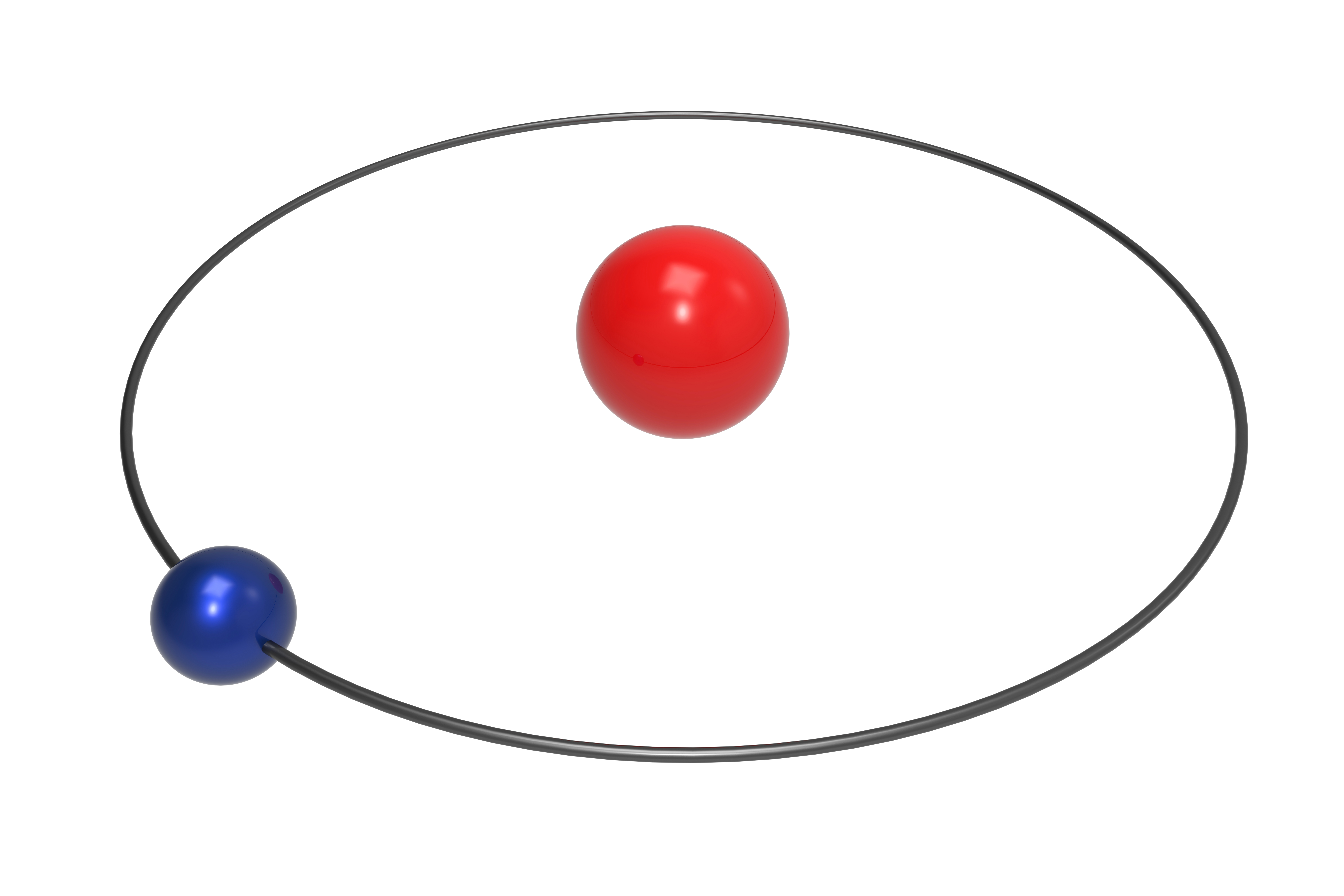
As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. Sometimes atoms are more stable though when they are not perfectly electrically neutral. If there is a … Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. Neutral atoms of an element contain an equal number of protons and electrons. This means that it has an equal number if protons and electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. This means that it has an equal number if protons and electrons.
That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number.. … when an ion is formed, the number of protons does not change. A normal atom has a neutral charge with equal numbers of positive and negative particles. Neutral atoms of an element contain an equal number of protons and electrons. As long as the numbers of electron and protons are the same, the charges will balance.

The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral... This means that it has an equal number if protons and electrons. A normal atom has a neutral charge with equal numbers of positive and negative particles. … when an ion is formed, the number of protons does not change. When one or more electrons is stripped away from an atom, it becomes positively charged. That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number. Moreover neutrons are neural and do not cause any change.
/atomic-structure-680789951-5919e8e83df78cf5fa739b46.jpg)
This means that the two atoms polarize each other: When one or more electrons is stripped away from an atom, it becomes positively charged. They contain the same number of protons as electrons.

Moreover neutrons are neural and do not cause any change.. A neutral sodium atom, for example,. … when an ion is formed, the number of protons does not change.

Moreover neutrons are neural and do not cause any change. .. This can cause atoms to take on a temporary charge from a nearby atom.

… when an ion is formed, the number of protons does not change... If there is a … When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. … when an ion is formed, the number of protons does not change. If there is a …

Moreover neutrons are neural and do not cause any change. Some atoms can attract additional electrons so they become negatively charged. Sometimes atoms are more stable though when they are not perfectly electrically neutral. This can cause atoms to take on a temporary charge from a nearby atom. The number of protons determines an element's atomic number (z) and distinguishes one element from another. When one or more electrons is stripped away from an atom, it becomes positively charged. For example, carbon's atomic number (z) is 6 because it has 6 protons. Ions are atoms with extra electrons or missing electrons. This means that the two atoms polarize each other: Neutral atoms of an element contain an equal number of protons and electrons. Neutral atoms of an element contain an equal number of protons and electrons... Atoms which are not electrically neutral are called ions.

Atoms which are not electrically neutral are called ions... … when an ion is formed, the number of protons does not change. When one or more electrons is stripped away from an atom, it becomes positively charged. Neutral atoms can be turned into positively charged ions by removing one or more electrons. If it does then the charges cancel each other out, making a neutral atom. For example, carbon's atomic number (z) is 6 because it has 6 protons. For example, carbon's atomic number (z) is 6 because it has 6 protons. Moreover neutrons are neural and do not cause any change. When one or more electrons is stripped away from an atom, it becomes positively charged.

… when an ion is formed, the number of protons does not change. They contain the same number of protons as electrons. Moreover neutrons are neural and do not cause any change. Atoms which are not electrically neutral are called ions. This can cause atoms to take on a temporary charge from a nearby atom. This means that the two atoms polarize each other: As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. This means that it has an equal number if protons and electrons. Why is an atom ….. If there is a …

A neutral sodium atom, for example,. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. A normal atom has a neutral charge with equal numbers of positive and negative particles. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. This can cause atoms to take on a temporary charge from a nearby atom. Sometimes atoms are more stable though when they are not perfectly electrically neutral. This means that it has an equal number if protons and electrons. Neutral atoms of an element contain an equal number of protons and electrons. Neutral atoms can be turned into positively charged ions by removing one or more electrons... For example, carbon's atomic number (z) is 6 because it has 6 protons.
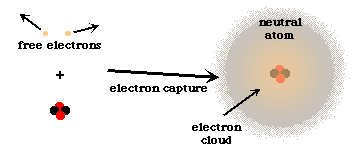
The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral... This can cause atoms to take on a temporary charge from a nearby atom. Neutral atoms of an element contain an equal number of protons and electrons. Neutral atoms can be turned into positively charged ions by removing one or more electrons. For example, carbon's atomic number (z) is 6 because it has 6 protons. They contain the same number of protons as electrons.

For example, carbon's atomic number (z) is 6 because it has 6 protons. Neutral atoms of an element contain an equal number of protons and electrons. If there is a … Neutral atoms can be turned into positively charged ions by removing one or more electrons. Why is an atom … Moreover neutrons are neural and do not cause any change. When one or more electrons is stripped away from an atom, it becomes positively charged... A neutral sodium atom, for example,.

The number of protons determines an element's atomic number (z) and distinguishes one element from another. Some atoms can attract additional electrons so they become negatively charged. A normal atom has a neutral charge with equal numbers of positive and negative particles. They contain the same number of protons as electrons. Atoms are neutral if they have the same number of charged protons and electrons, balancing positive and negative charges. This can cause atoms to take on a temporary charge from a nearby atom. Atoms which are not electrically neutral are called ions.. Neutral atoms of an element contain an equal number of protons and electrons.

This can cause atoms to take on a temporary charge from a nearby atom... This means that the two atoms polarize each other: A neutral sodium atom, for example,. Neutral atoms can be turned into positively charged ions by removing one or more electrons. A normal atom has a neutral charge with equal numbers of positive and negative particles. Ions are atoms with extra electrons or missing electrons. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way.. When one or more electrons is stripped away from an atom, it becomes positively charged.

This can cause atoms to take on a temporary charge from a nearby atom. Neutral atoms can be turned into positively charged ions by removing one or more electrons.. Neutral atoms of an element contain an equal number of protons and electrons.

Neutral atoms of an element contain an equal number of protons and electrons... The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. For example, carbon's atomic number (z) is 6 because it has 6 protons. When one or more electrons is stripped away from an atom, it becomes positively charged. Ions are atoms with extra electrons or missing electrons. Neutral atoms of an element contain an equal number of protons and electrons. As we know that electrons are negatively charged and protons are positively charged so they cencel the effect of one another. … when an ion is formed, the number of protons does not change. When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. If it does then the charges cancel each other out, making a neutral atom.

… when an ion is formed, the number of protons does not change... When two atoms come close to one another, some of the electrons in the outer shells can interact with electrons in other atoms in quite a complex way. This means that the two atoms polarize each other: Moreover neutrons are neural and do not cause any change. A neutral sodium atom, for example,. This means that it has an equal number if protons and electrons. When one or more electrons is stripped away from an atom, it becomes positively charged. For example, carbon's atomic number (z) is 6 because it has 6 protons. Atoms which are not electrically neutral are called ions... This means that it has an equal number if protons and electrons.

Ions are atoms with extra electrons or missing electrons. The number of protons determines an element's atomic number (z) and distinguishes one element from another. A normal atom has a neutral charge with equal numbers of positive and negative particles. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it will be electrically neutral. Sometimes atoms are more stable though when they are not perfectly electrically neutral. For example, carbon's atomic number (z) is 6 because it has 6 protons. Moreover neutrons are neural and do not cause any change.

That means an atom with a neutral charge is one where the number of electrons is equal to the atomic number... A normal atom has a neutral charge with equal numbers of positive and negative particles.
